Pengaruh Variasi Dopan Asam Terhadap Kinerja Baterai Sekunder Polianilina|Zn
Downloads
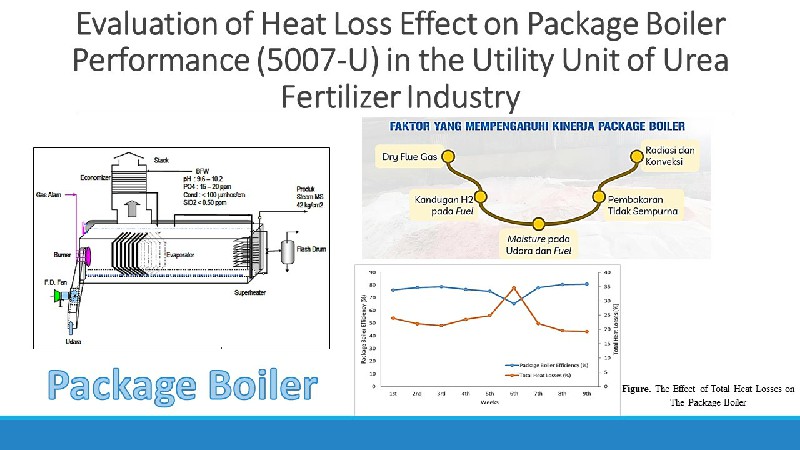
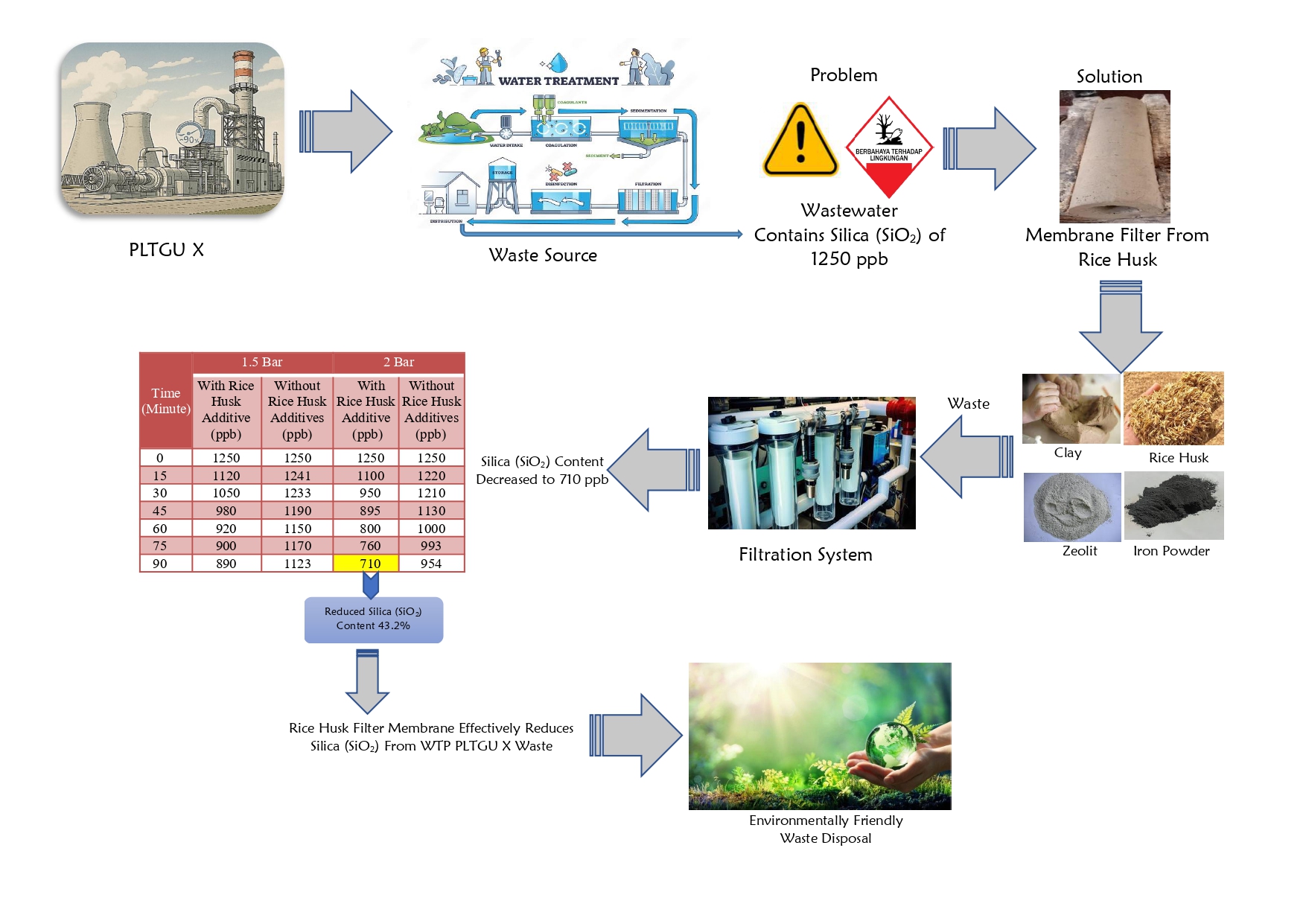
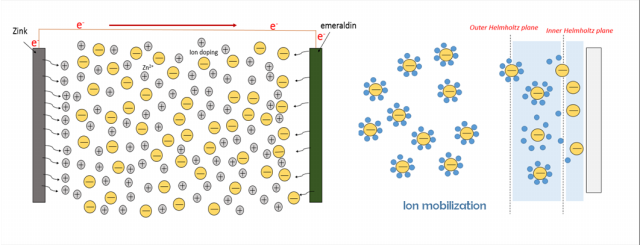
Studi tentang baterai sekunder polianilina(PAni)|Zn telah banyak dikembangkan, tetapi sejauh ini masih belum ada penjelasan mengenai pengaruh asam terhadap kinerja baterai tersebut. PAni disitensis menggunakan metoda elektrodeposisi dengan tegangan 0,7 V selama 30 menit. PAni hasil sintesis dikarakterisasi menggunakan Voltammetri dan Spektroskopi Inframerah. Baterai PAni|Zn didesain dalam bentuk sandwich, kemudian pengukuran kinerja digunakan impedansi serta pengisian-pengosongan. Baterai dengan elektroda PAni-Cl dan PAni-Br mempunyai kinerja yang lebih baik dengan specific capacity saat ke-60 yaitu 55,4 dan 37,4 mAh g-1. Pengukuran impedansi pada baterai dengan elektroda PAni-Cl, PAni-Br, dan PAni HClO4, menunjukan resistance solution (Rs) secara berurutan yaitu 1,38; 2,56; dan 3,03 W dan resistance charge transfer (Rct) 2,24; 2,97; dan 7,71 W. Oleh sebab itu, baterai PAni|Zn dengan dopan HCl menunjukkan kinerja terbaik dibanding dengan asam yang lain.
Downloads
Chang, X., Hu, R., Sun, S., Liu, J., Lei, Y., Liu, T., Dong, L., & Yin, Y. (2018). Sunlight-charged electrochromic battery based on hybrid film of tungsten oxide and polyaniline. Applied Surface Science, 441, 105-112. https://doi.org/10.1016/j.apsusc.2018.02.003
Chen, Y., & Manzhos, S. (2016). Voltage and capacity control of polyaniline based organic cathodes: An ab initio study. Journal of Power Sources, 336, 126-131. https://doi.org/10.1016/j.jpowsour.2016.10.066
Deyab, M. A., & Mele, G. (2019). Polyaniline/Zn-phthalocyanines nanocomposite for protecting zinc electrode in Zn-air battery. Journal of Power Sources, 443, 227264. https://doi.org/10.1016/j.jpowsour.2019.227264
Gao, H., Lu, Q., Yao, Y., Wang, X., & Wang, F. (2017). Significantly Raising the Cell Performance of Lithium Sulfur Battery via the Multifunctional Polyaniline Binder. Electrochimica Acta, 232, 414-421. https://doi.org/10.1016/j.electacta.2017.02.160
Hatchett, D. W., Josowicz, M., & Janata, J. (1999). Acid doping of poly aniline: Spectroscopic and electrochemical studies. Journal of Physical Chemistry B, 103(50), 10992-10998. https://doi.org/10.1021/jp991110z
Huang, J., Tu, J., Lv, Y., Liu, Y., Huang, H., Li, L., & Yao, J. (2020). Achieving mesoporous MnO2@polyaniline nanohybrids via a gas/liquid interfacial reaction between aniline and KMnO4 aqueous solution towards Zn-MnO2 battery. Synthetic Metals, 266, 116438. https://doi.org/10.1016/j.synthmet.2020.116438
Kawashima, H., Okatani, R., Mayama, H., Nakamura, Y., & Fujii, S. (2018). Synthesis of hydrophobic polyanilines as a light-responsive liquid marble stabilizer. Polymer, 148, 217-227. https://doi.org/10.1016/j.polymer.2018.06.039
Kurada, K. V., & De, S. (2018). Polyaniline doped ultrafiltration membranes: Mechanism of membrane formation and pH response characteristics. Polymer, 153, 201-213. https://doi.org/10.1016/j.polymer.2018.08.032
Li, X., Lv, R., Zou, S., Na, B., Liu, P., Ma, Y., & Liu, H. (2019). Polyaniline nanopillars on surface cracked carbon fibers as an ultrahigh-performance cathode for a flexible rechargeable aqueous Zn-ion battery. Composites Science and Technology, 180, 71-77. https://doi.org/10.1016/j.compscitech.2019.05.016
Luo, J., Zhong, W., Zou, Y., Xiong, C., & Yang, W. (2016). Preparation of morphology-controllable polyaniline and polyaniline/graphene hydrogels for high performance binder-free supercapacitor electrodes. Journal of Power Sources, 319, 73-81. https://doi.org/10.1016/j.jpowsour.2016.04.004
Copyright (c) 2020 CHEESA: Chemical Engineering Research Articles

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
With the receipt of the article by CHEESA Editorial Board and the decision to be published, the copyright regarding the article will be transferred to CHEESA Journal.
CHEESA has the right to multiply and distribute the article and every author is not allowed to publish the same article that was published in this journal.

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Under the following terms:
Attribution ” You must give appropriate credit, provide a link to the license, and indicate if changes were made. You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use.
NonCommercial ” You may not use the material for commercial purposes.
ShareAlike ” If you remix, transform, or build upon the material, you must distribute your contributions under the same license as the original.